
Waterproof and Breathable Wound Care Dressing Wound Hydrocolloid Dressing for Knife Wounds, Minor Bruises, Surgical Wounds, Ulcers Pressure Sores
Basic Info
| Advantage | Closed Wound, Debridement Function |
| Product Type | Back/Thin |
| Piece/Box | 10 |
| PCS./Box | 100 |
| Model | Non Self-Adhesive/Self-Adhesive Type |
| Transport Package | Box |
| Trademark | TECHDRIVE |
| Origin | Guangdong, Zhongshan |
| HS Code | 3005109000 |
| Production Capacity | 100000 Pieces/Week |
Product Description
Waterproof and Breathable Wound Care Dressing Wound Hydrocolloid Dressing for Knife wounds,Minor bruises,Surgical wounds,Ulcers Pressure sores

Product Description
The hydrocolloid dressing combines the water absorption properties of the water -soluble polymer and the viscosity of the rubber material. The wet healing environment supplied by hydrocolloid dressing has a great effect on promoting wound healing, which can fully absorb wound exudate and prevent bacteria from invading wounds.
Intended UseKnife woundsMinor bruisesSurgical woundsUlcers Pressure sores
Features
Mechanism
Moist Balance
Easy Observation
Foot injuries from shoe fiction
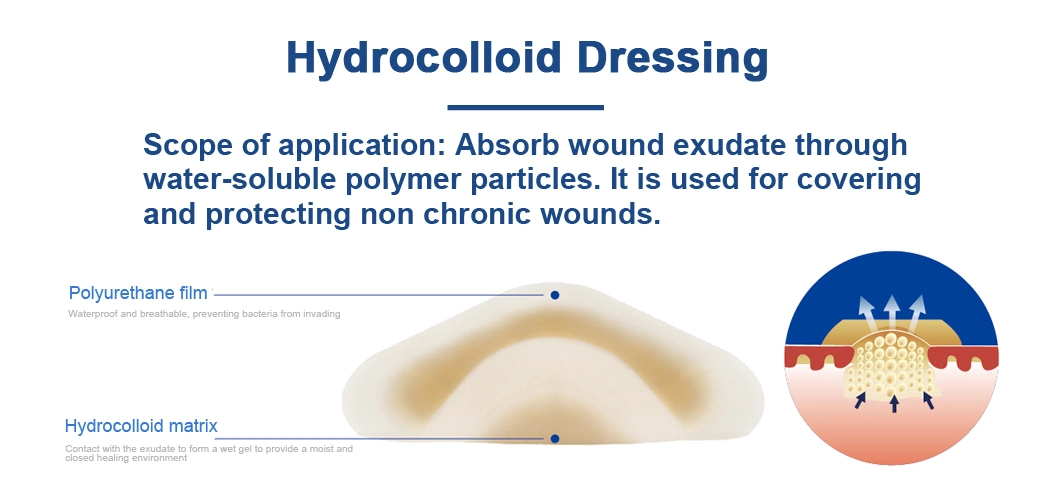
Product Parameters
| model | Specifications | Piece/box | Pcs./box |
| Non self-adhesive | 5cm*5cm | 10 | 100 |
| *10cm*10cm | 10 | 100100100 | |
| 10cm*20cm | 10 | ||
| *2cm*30cm | 10 | ||
| Self-adhesive type | 6cm*7cm | 10 | 100 |
| 7cm*9cm | 10 | 100 |
| Type | Code | Size(cm) |
| Regular | 060518 | 5x1.8 |
| 060602 | 6x2 | |
| 064628 | 4.6x2.8 | |
| Premium | 071010 | 10x10 |
| 071515 | 15x15 | |
| 072020 | 20x20 |
Detailed Photos
Installation Instructions
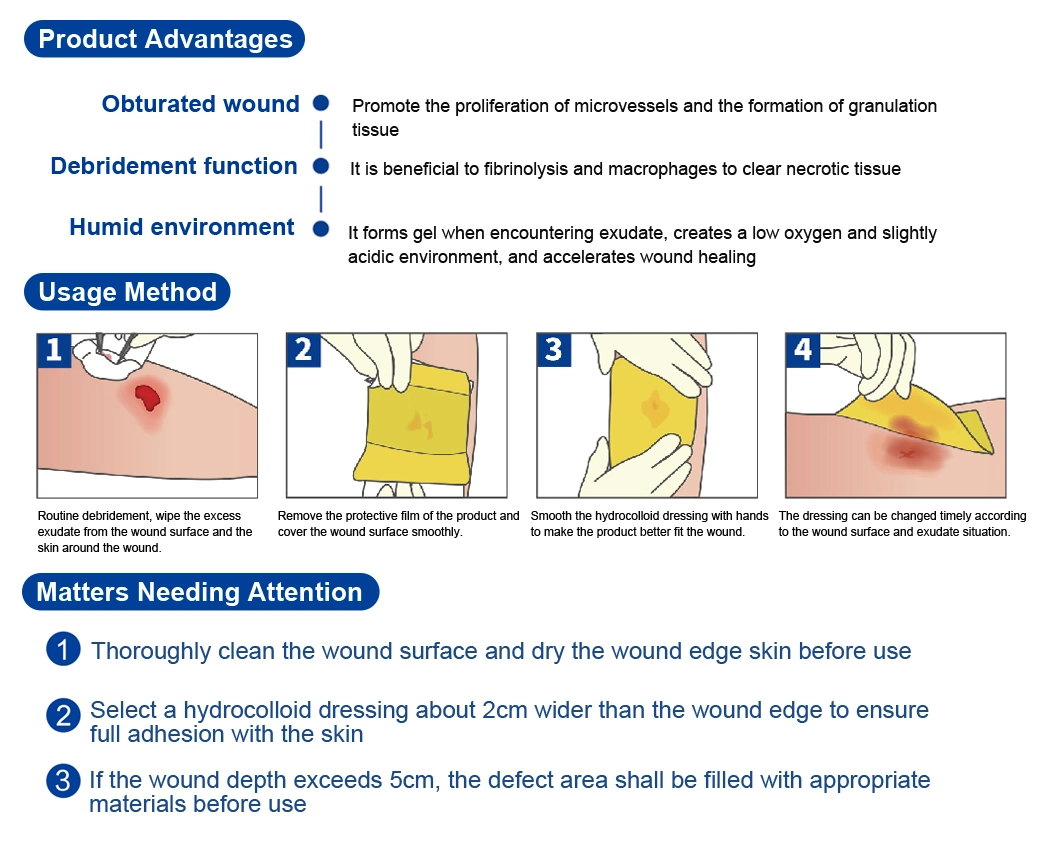
Our Advantages
Closed wound: promote the proliferation of microvessels and the formation of granulation tissueDebridement function: it is beneficial to fibrinolysis and macrophages to remove necrotic tissueHumid environment: form gel when encountering exudate, create a low oxygen and slightly acidic environment, and accelerate wound healing
Company Profile
Guangdong Tidakang Medical Technology Is a medical technology company approved by Guangdong Provincial People's government and registered by Guangdong Food and Drug Administration. Founded in 2015, with a registered capital of 10 million yuan, the company invested more than 6 million yuan to introduce advanced production equipment from abroad at the beginning of its establishment, and has a perfect and scientific management system. After two years of development, it has become a rising star in the industry. The company has its own ability to develop, produce and sell medical surface catheter fixation devices, transparent stickers, transparent dressings, fixation bands and other products. The company has the production qualification of Class I and Class II medical devices. We welcome people from all walks of life to visit our company.Exhibition photos
Certifications






